 As proof of concept, siRNA against those genes along with a GAPDH (typically thought of as a housekeeping gene) control was used. An RNA affinity chromatography pulldown identified hnRNPs (heterogeneous ribonucleoproteins), glutamyl-prolyl tRNA synthetase (EPRS) and poly (A)-binding protein (PABP) as candidates. 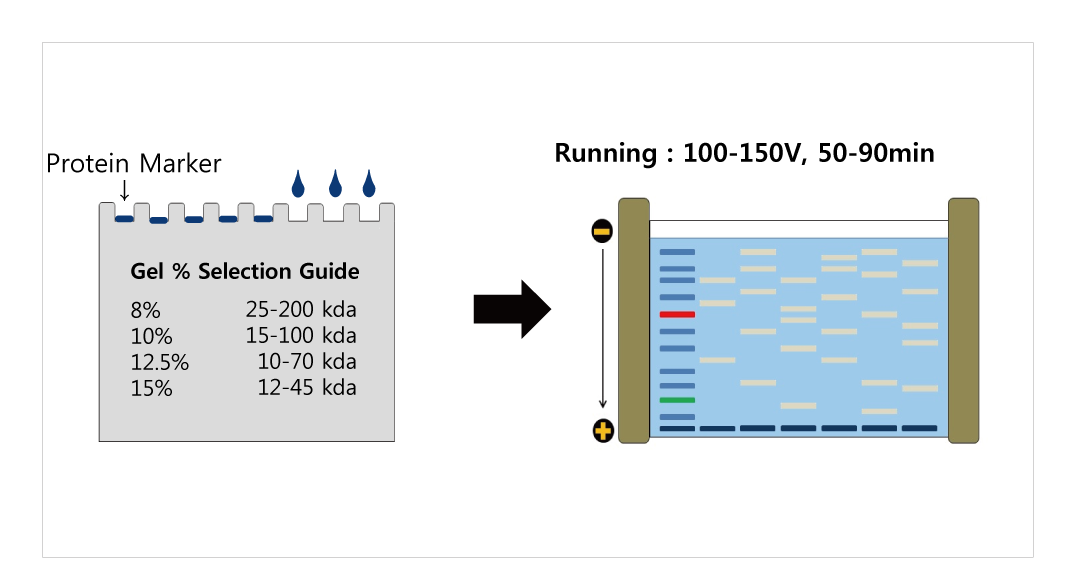
Yet, prior to these studies, the inhibitory role of GAPDH against coronavirus was demonstrated in a study that used the transmissible gastroenteritis coronavirus to help identify proteins required for RNA transcription of the coronavirus virome ( 11). By examining different coronavirus studies, this review explores GAPDH's role as an inhibitor of coronaviruses (at the viral transcriptional level and also as a modulator of gene expression related to inflammation), and its signal transduction links to the IFNγ and NO pathways.Ī number of recent studies have focused on the examination of host proteins in terms of their potential pro- and antiviral properties against coronaviruses ( 1– 9). These observations would imply that sufficient levels of GAPDH are needed for immune responses to function properly during a coronaviral infection. Lastly, another important role for GAPDH is its requirement in the assembly of the GAIT complex that is responsible for termination of translation of IFNγ-responsive genes that would be critical for the resolution of any inflammatory response. Additionally, GAPDH has also been shown to be a negative transcriptional regulator of AT1R (angiotensin II receptor 1), which has been shown to bind ACE2 for eventual endocytosis of the complex implicating GAPDH's potential role in the kinetics of coronavirus entry as well in downstream inflammatory signaling resulting from AT1R activation. Although perhaps better known for its role in glycolysis, GAPDH also plays a role in gene expression of a varied set of genes by binding to their mRNA to affect stability and thereby translation Moreover, GAPDH is also upregulated by nitric oxide (NO), an inhibitor against both SARS-CoV and SARS-CoV-2. In this light, the previous coronavirus strain (SARS-CoV) has actually been shown to downregulate GAPDH. Studies have show that downregulation of GAPDH leads to a decrease in interferon gamma (IFNγ production (which is an important cytokine response against coronaviruses and viruses in general). One possible (and perhaps unlikely) therapeutic target could be GAPDH (glyceraldehyde-3-phosphate dehydrogenase). However, this pandemic presents a unique opportunity to investigate anti-viral therapies given the likely probability of another outbreak. Independent Researcher, Princeton, NJ, United StatesĪs the COVID-19 pandemic finishes its second year, progress has been made against SARS-CoV-2 with vaccine candidates showing efficacy against this latest coronavirus strain.SNAP23 is essential for platelet and mast cell development and required in connective tissue mast cells for anaphylaxis. A mouse model of BBS identifies developmental and homeostatic effects of BBS5 mutation and identifies novel pituitary abnormalities. Aneuploidy renders cancer cells vulnerable to mitotic checkpoint inhibition. PHLDA1 knockdown inhibits inflammation and oxidative stress by regulating JNK/ERK pathway, and plays a protective role in sepsis-induced acute kidney injury. 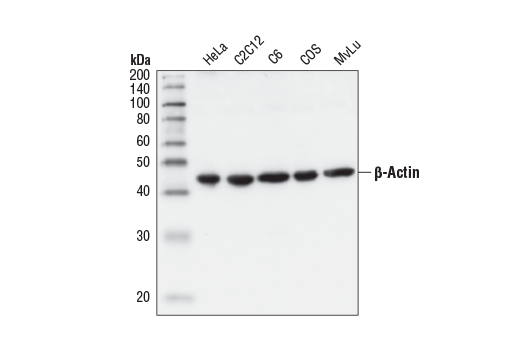
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
